Pathophysiology of COVID-19-associated coagulopathy and its impact on laboratory measures of coagulation
Severe Acute Respiratory Syndrome Coronavirus – 2 (SARS-CoV-2) is the virus that causes COVID-19, which has ravaged the world since January 2020, infecting and killing millions of people worldwide. Based on the global numbers of the total number of individuals infected, approximately 2 percent of confirmed SARS-CoV-2 infections succumb to the disease. The infection may be asymptomatic or may cause a broad range of symptoms including, but not limited to, mild symptoms of the upper respiratory tract, cytokine storm, multi-organ failure, life-threatening sepsis, and COVID-19-associated coagulopathy, which causes serious thrombotic complications. The exploration and understanding of the mechanisms behind the severe course of COVID-19 is crucial to improve treatment, recovery, and ultimately the survival rate of hospitalized patients. Laboratory parameters are not only essential to better understand the physiological mechanisms of the disease, but also to monitor disease progression and evaluate the effectiveness and success of treatment.
The cytokine storm
An early sign of severe COVID-19, and a predictor for worse outcomes, is the occurrence of the cytokine storm. A cytokine storm is an unregulated and excessive release of pro-inflammatory cytokines, first locally in the infected lung and later, systemic throughout the body.1 High plasma levels of inflammatory markers [C-reactive protein (CRP), serum amyloid A (SAA), ferritin, procalcitonin] and cytokines [Interleukin-6 (IL-6), Interleukin-10 (IL-10) and Tumor Necrosis Factor-α (TNF-α)] are indicators of a hyperinflammatory response and an underlying cytokine storm.2,3 The increase in pro-inflammatory cytokines (in particular, IL-6, TNF- α, and IL-10), which are elevated in patients with COVID-19, are indicators of a cytokine storm and are associated with disease progression and outcomes in severe COVID-19 patients.4,5 Currently, it is unclear if immune hyperactivity, dysregulation of the inflammatory response to the viral infection, or immune dysregulation causes the progression to severe COVID-19.1
COVID-19-associated coagulopathy
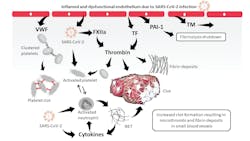
The cytokine storm, with its excessive release of pro-inflammatory cytokines, may also play a key role in the pathophysiology of COVID-19-associated coagulopathy by activating endothelial cells and leukocytes – in particular, neutrophils – which in response, produce neutrophil extracellular traps (NETs), a process called NETosis. NETs promote thrombus formation, amplify cytokine production, and play a significant role in the pathophysiology of COVID-19.6 In addition to the activation of endothelial cells and neutrophils, the direct interaction of the virus with the contact system, part of the innate immune system – specifically with that of factor XII (FXII) and plasma prekallikrein – may also contribute to the highly prothrombotic environment at the site of infection.7 The formation of activated FXII via SARS-CoV-2 contact may not only initiate thrombosis via the intrinsic pathway of coagulation [e.g., increase thrombin generation, fibrin formation (microthrombosis), fibrinolysis, and increased D-dimer levels], but also the production of bradykinin, which increases vascular dilation and permeability. Thus, SARS-CoV-2 directly and indirectly influences the coagulation system, creating a highly prothrombotic state in patients with COVID-19. Figure 1 schematically summarizes the pathophysiological mechanisms of COVID-19-associated coagulopathy.
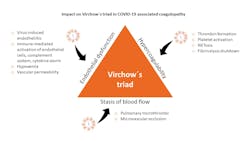
The more than a century-old concept of Virchow`s triad shows how three factors contribute to thromboembolic risk: endothelial dysfunction, blood flow/stasis, and hypercoagulability. The three factors highlighted by Virchow can explain the venous and arterial hypercoagulable state seen in COVID-19 patients (Figure 2).8
In summary, thrombosis in COVID-19 patients is a complex and multifactorial process, and COVID-19-associated coagulopathy must be considered to successfully treat and prevent potentially lethal thrombotic complications.
Thrombosis in COVID-19
While it is not unusual for infections to raise the risk of thrombosis, the highly prothrombotic state in COVID-19 patients causes an unprecedented range of thrombosis-related disorders in affected patients. From benign skin lesions on the feet (e.g., COVID toe), to life-threatening thrombotic events, the SARS-CoV-2 virus has demonstrated a strikingly high prevalence of deadly blood clots. Early studies have shown that approximately 25-45 percent9-11 — or even up to 70 percent12 — of critically ill patients have a confirmed venous thromboembolism (VTE), such as deep vein thrombosis (DVT) or pulmonary embolism (PE). Approximately 70 percent of COVID-19 patients who died had met the International Society of Thrombosis and Hemostasis (ISTH) criteria for disseminated intravascular coagulation (DIC).13 Analyzing available study data, the weighted mean prevalence of VTE was found to be as high as 31.3 percent in COVID-19 patients,11 while the pooled incidence of VTE in COVID-19 patients admitted to the ICU was 28 percent.14 The incidence of VTE can steadily increase in hospitalized patients during severe COVID-19, from 16 percent after 1 week to 42 percent after 3 weeks.15 When comparing those findings with the incidence of VTE in patients with pneumonia following respiratory tract infections, the rate of VTE in COVID-19 patients is 7- to 8-fold higher.11
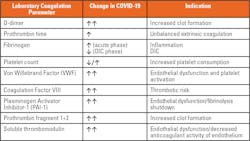
An autopsy on 12 deceased COVID-19 patients revealed DVT in 7 of 12 patients (58 percent), while PE was the direct cause of death in 4 patients (33 percent).16 A histological analysis of pulmonary vessels in COVID-19 patients showed thrombosis and endotheliitis throughout the pulmonary vasculature, with alveolar capillary microthrombi being ninefold more prevalent in COVID-19 patients than in patients with H1N1 influenza.17 The high incidence of thromboembolic events in COVID-19 patients, which are also frequently the cause of death, highlight the importance of diagnosing and treating COVID-19-associated coagulopathy. One of the most relevant laboratory parameters for diagnosing and monitoring COVID-19-associated coagulopathy is the determination of plasma D-dimer levels. Other laboratory parameters of coagulation are also altered in COVID-19 patients, as described in Table 1.
Following a COVID-19 diagnosis, hemostasis testing, monitoring, and therapy have been shown to play a decisive role in COVID-19 patient management.
D-dimer – a primary marker for COVID-19-associated coagulopathy
Elevated D-dimer levels were found to be a crucial laboratory marker to indicate a thrombotic risk in COVID-19 patients.18,19 However, when interpreting D-dimer results, several aspects must be considered. For example, D-dimer levels can increase with age and are elevated during pregnancy. 20 In addition, high plasma D-dimer levels are also observed in a variety of clinical conditions including but not limited to DIC, sepsis, inflammation, DVT/PE, immobility, liver disease, malignancy, recent surgery, preeclampsia, and trauma. D-dimer is primarily used to rule out DVT/PE in low risk-patients (non-hospitalized), due to its high negative predictive value, and to diagnose and monitor DIC in conjunction with other laboratory parameters. It is also important to note that D-dimer is not a clearly defined antigen. It consists of multiple D/E-fragments of different molecular weights. Thus, the D-dimer antigen is heterogenous, as are the antibodies used to measure it. As a result, the D-dimer assays cannot yet be standardized to an international standard. Each assay uses its own calibration material, which means comparing or transferring D-dimer results has to be done with great caution. Some assays use fibrinogen equivalent units (FEU), while other assays report in D-dimer units (DDU). D-dimer results are more commonly reported in FEUs (FEU = 2xDDU).
One of the first reports highlighting the importance of D-Dimer was a study of 191 hospitalized COVID-19 patients in Wuhan (137 survivors and 54 non-survivors), which identified elevated D-dimers levels greater than 1 µg/mL FEU (odds ratio: 18.42, 2.64–128.55; p=0·0033) on admission as a strong predictor of in-hospital death.21 Another retrospective study from Wuhan, China, looked at 343 COVID-19 patients, of whom 330 survived and 13 died.22 A D-dimer cutoff of 2 µg/mL was derived by receiver operator characteristics (ROC) curve analysis which yielded 92.3 percent sensitivity and 83.3 percent specificity in predicting in-hospital mortality. In addition, using Kaplan-Meier curves, patients with COVID-19 with D-dimer levels >2 μg/mL FEU on admission were shown to be 50 times more likely to die than patients with D-dimer levels <2 μg/mL FEU.
Comparable results regarding the D-dimer cutoff and its predictive power were found in a study on 248 hospitalized COVID-19 patients with 17 non-survivors.23 ROC curve analysis revealed a cutoff of >2.14 µg/mL FEU of D-dimer on admission to predict death, with an odds ratio (OR) of 10.17 (95 percent CI 1.10-94.38, P=0.041).
Another multicenter retrospective study that included 400 hospitalized patients showed that an elevated D-dimer >2.5 µg/mL at initial presentation was predictive of coagulation-associated complications during hospitalization, including thrombosis, bleeding, critical illness, and death.19
As increased D-dimer levels are common in COVID-19, the exclusion or diagnosis of VTE using plasma D-dimer may need to be adapted. The widely used cut-off for the exclusion of VTE in non-COVID-19 patients of <0.5 µg/mL FEU is not suitable. A study analyzing VTE in 100 COVID-19 patients admitted to the intensive care unit (ICU) calculated an optimal cutoff for the exclusion of VTE of <2.0 µg/mL applying ROC curve analysis to their data set.24 However, a cutoff value of >8.0 µg/mL provided an optimal sensitivity and specificity for the diagnosis of VTE in COVID-19 patients. The authors conclude that those two D-dimer cutoffs may be useful to identify patients with a low or high probability for the presence of VTE.
The above studies indicate that D-dimer can be used as an early prognostic marker for COVID-19 progression, including mortality and thrombotic complications, as well as a helpful marker to improve management and outcomes in COVID-19 patients.
Other markers of COVID-19-associated coagulopathy
Although D-dimer has been used extensively as a laboratory marker in COVID-19, other laboratory markers of coagulation are also altered.25 For example, prothrombin fragment 1+2, an early indicator of thrombin formation, was found to be markedly increased in COVID-19 patients.26 In addition, fibrinogen, von Willebrand Factor (VWF) and factor VIII (FVIII), hemostasis-associated acute-phase reactant proteins, are also elevated in COVID-19 (Table 1).25,27 Fibrinogen levels are increased in hospitalized patients due to hyperinflammatory response.19 However, early studies have shown a markedly reduced fibrinogen mimicking levels associated with DIC in patients with severe COVID-19.13 The significant increase in FVIII and VWF not only confer an increase in thrombotic risk in COVID-19 patients, but are also markers of endothelial activation.25
High plasma levels of VWF promote tethering of platelets to the inflamed endothelium, which may lead to platelet activation as shown by increased P-selectin expression of platelets isolated from COVID-19 patients (Fig. 1).28 Platelets from severe COVID-19 patients were assessed under high shear conditions using the PFA-200 system, which showed decreased closure times compared to those of patients with intermediate COVID-19, indicating that platelets of patients with severe COVID-19 are hyperactive. 28
Increased plasma levels of soluble thrombomodulin and plasminogen activator inhibitor-1 (PAI-1) have also been reported.25 Only thrombomodulin bound to the extracellular membrane of endothelial cells can convert activated thrombin (FIIa) from being prothrombotic to being antithrombotic. An increase in soluble thrombomodulin means a decrease in membrane-bound thrombomodulin and its ability to attenuate clot formation. PAI-1 inhibits fibrinolysis, the endogenous process of resolving blood clots. However, under high concentrations, as seen in inflammatory responses, PAI-1 can tip the balance of coagulation in the direction of thrombosis.
Furthermore, Nicolai and colleagues28 evaluated the formation of neutrophil extracellular traps, or NETosis, in patients with COVID-19. Conducting an experiment in which platelet-rich plasma isolated from either healthy donors or COVID-19 patients was incubated with control neutrophils, and analyzing the formation of NETs by confocal microscopy, an enhanced NETosis was noticed in severe COVID-19 patients.
The alteration of all those coagulation parameters in plasma are pointing in the same direction: COVID-19-associated coagulopathy is a hypercoagulable and highly prothrombotic state requiring thorough treatment and monitoring.
Therapy and monitoring of COVID-19-associated coagulopathy
Various societies and expert groups have issued treatment and monitoring recommendations for COVID-19-associated coagulopathy.29-33 Daily measurements of D-dimer and fibrinogen levels, prothrombin time (PT), and platelet count are indicated to detect and monitor COVID-19-associated coagulopathy, as well as the success of anticoagulant therapy. Anticoagulation with prophylactic to therapeutic doses of preferably low-molecular weight heparins (LMWHs), or unfractionated heparins (UFH), is strongly recommended. Dosing depends on disease status and risk profile of the patient.29-32 Therapeutic monitoring of the heparins using anti-FXa-assays – but not the APTT (activated partial thromboplastin time) – is preferred. Anticoagulant therapy success may be monitored using plasma D-dimer levels.
Several clinical trials are currently underway to help optimize treatment protocols from drug dosing, application timing, and the choice of the right anticoagulant drug(s) and other potentially helpful drugs, such as the antiplatelet drug clopidogrel, tissue plasminogen activator (tPA), thrombomodulin, and antithrombin. Anticoagulation predominantly with LMWH is now standard of care for hospitalized COVID-19 patients. But the high incidence of thrombotic complications in COVID-19, despite anticoagulation with LMWH, clearly shows that there is still a lot room for improvement in treating and managing COVID-19-associated coagulopathy.